Recently, Associate Professor Wang Shengjie from Kangda College and the Lianyungang Medical Education & Innovation Research Center at Nanjing Medical University (NMU), in collaboration with Professor Zhang Hongtao from Soochow University and Associate Professor Jin Ersuo from Jiaxing University, published a research paper titled“STAT3-mediated transactivation of NOVA2 promotes lung adenocarcinoma metastasis by splicing SMAD4” in Oncogene,an internationally renowned journal.
This study systematically reveals the crucial role of the RNA-binding protein NOVA2 in lung adenocarcinoma metastasis and its epigenetic–transcriptional–splicing cascade regulatory mechanism, providing a new potential target for precision diagnosis and treatment of lung adenocarcinoma. The paper lists Kangda College of NMU and the Lianyungang Medical Education & Innovation Research Center of NMU as theprimary and corresponding affiliation.

Lung cancer is the leading cause of both incidence and mortalityworldwide, with lung adenocarcinoma being the most prevalent histological subtype. Metastasis is the primary cause of death among these patients. Although the role of the TGF-β/SMAD signaling pathway in tumor progression has been widely recognized, the mechanisms by which RNA-binding proteins influence this pathway via alternative splicing to drive lung adenocarcinoma metastasis remain unclear.
By integrating bioinformatics analysis with large-scale clinical sample data, the research team identified NOVA2, an RNA-binding protein, as significantly overexpressed in metastatic lung adenocarcinoma. They found that the NOVA2 expression level was closely associated with poor prognosis and lymph node metastasis in patients with lung adenocarcinoma. Knockdown of NOVA2 suppressed epithelial–mesenchymal transition (EMT) and invasive metastasis of lung adenocarcinoma cells.
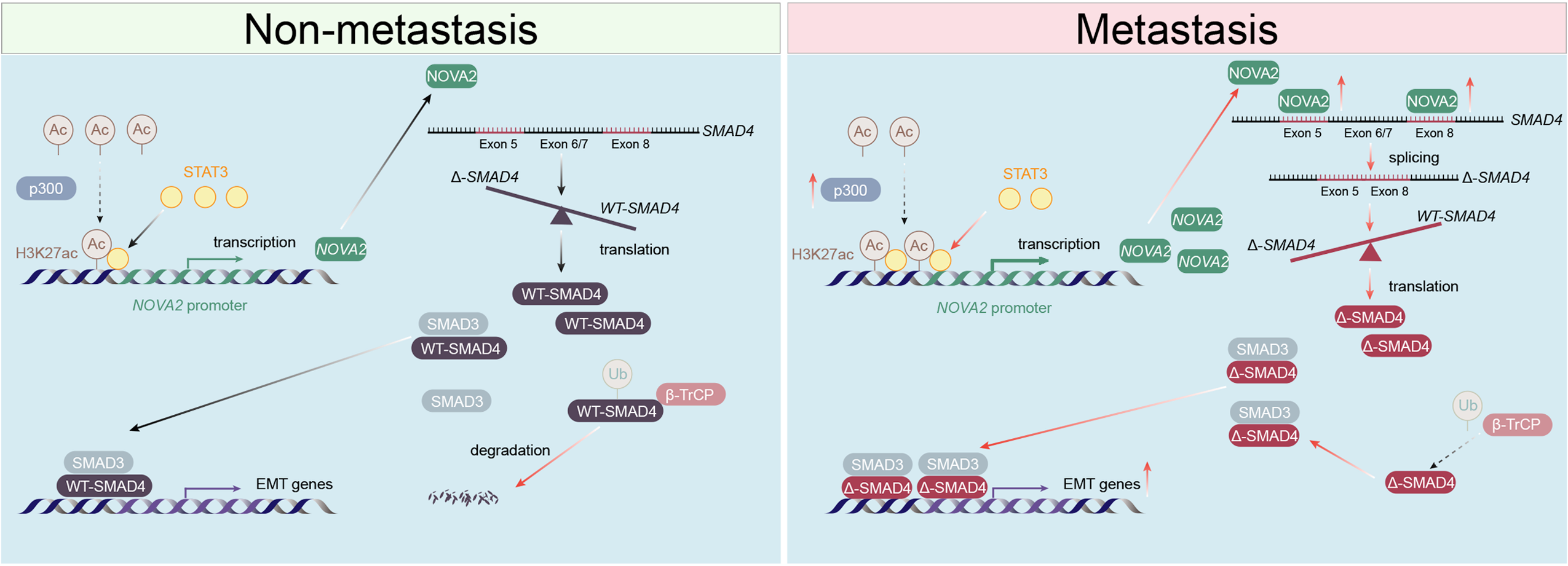
At the molecular level, the study further revealed that enhanced H3K27 acetylation, mediated by the histone acetyltransferase p300, promotes the binding of transcription factor STAT3 to the NOVA2 promoter region, and subsequent transcriptional activation of NOVA2. The upregulated NOVA2 then regulates the alternative splicing of the SMAD4 gene, leading to the generation of a truncated isoform, Δ-SMAD4, which lacks exons 6 and 7. This isoform escapes β-TrCP-mediated ubiquitination degradation, thereby becoming more stable, cotinuously activating the TGF-β/SMAD signaling pathway, and ultimately driving EMT and metastasis in lung adenocarcinoma cells.
This study offers a comprehensive understanding of the STAT3/NOVA2/Δ-SMAD4 molecular axis in lung adenocarcinoma metastasis across multiple levels—epigenetic regulation, transcriptional activation, alternative splicing, and signaling pathways (see figure below). These findings deepen our understanding of the molecular basis of lung adenocarcinoma metastasis and provides a new theoretical foundation for developing targeted therapeutic strategies against this pathway.
The co-corresponding authors of this paper are Associate Professor Wang Shengjie from Kangda College and Lianyungang Medical Education & Innovation Research Center, NMU, Professor Zhang Hongtao from Soochow University, and Associate Professor Jin Ersuo from Jiaxing University The co-first authors include Associate Professor Wang Shengjie, along with Assistant Researcher Tong Xin from the First Affiliated Hospital of Soochow University and Director Sun Runfeng from Donghai County People’s Hospital.
This work was supported by grants from the National Natural Science Foundation of China, the Natural Science Foundation of Jiangsu Province, and the Key Project (Category A) of the NMU Lianyungang Medical Education & Innovation Research Center.
Link to original article:https://www.nature.com/articles/s41388-026-03752-6
(Drafted by Kangda College of Nanjing Medical University; Reviewed by Haifeng Wang; Translation revised by Bei Zhang)



