Recently, the research team of Li Kai and Han Xiao from the School of Basic Medical Sciences at Nanjing Medical University (NMU) published an online research paper in Cell Reports titled “Ets1-dependent Mek-Erk signaling drives adipose tissue macrophage anti-inflammatory polarization to ameliorates insulin resistance”. This study identifies Ets1, a transcription factor, as a key effector in the Mek-Erk signaling pathway, which specifically responds to IL-4 signaling to drive macrophage polarization toward an anti-inflammatory M2 phenotype, thereby alleviating adipose tissue inflammation and insulin resistance.
Obesity has become a significant global public health challenge, withchronic low-grade inflammation in adipose tissue being a key driver of insulin resistance and type 2 diabetes. Adipose tissue macrophages (ATMs) are the most abundant immune cells in adipose tissue. In obesity, their phenotype polarizes from the anti-inflammatory M2 type to the pro-inflammatory M1 type, exacerbating metabolic disorders. Therefore, modulating the M2/M1 balance of ATMs is considered a promising strategy for improving obesity-related insulin resistance.
The M2/M1 polarization of macrophages is tightly regulated by multiple signaling pathways. Among them, the Mek-Erk pathway of the mitogen-activated protein kinase (MAPK) family plays a complex and paradoxical role.On the one hand, M2 polarization requires Mek-Erk activity; on the other hand, pro-inflammatory stimuli (such as LPS) also activate Erk. Clinical studies have found that the use of MEK/ERK inhibitors (e.g., trametinib, cobimetinib) showsopposing immunomodulatory effects across different studies. This functional divergence aries from the diversity of Erkdownstream effectors, but the underlying mechanisms remain unclear.
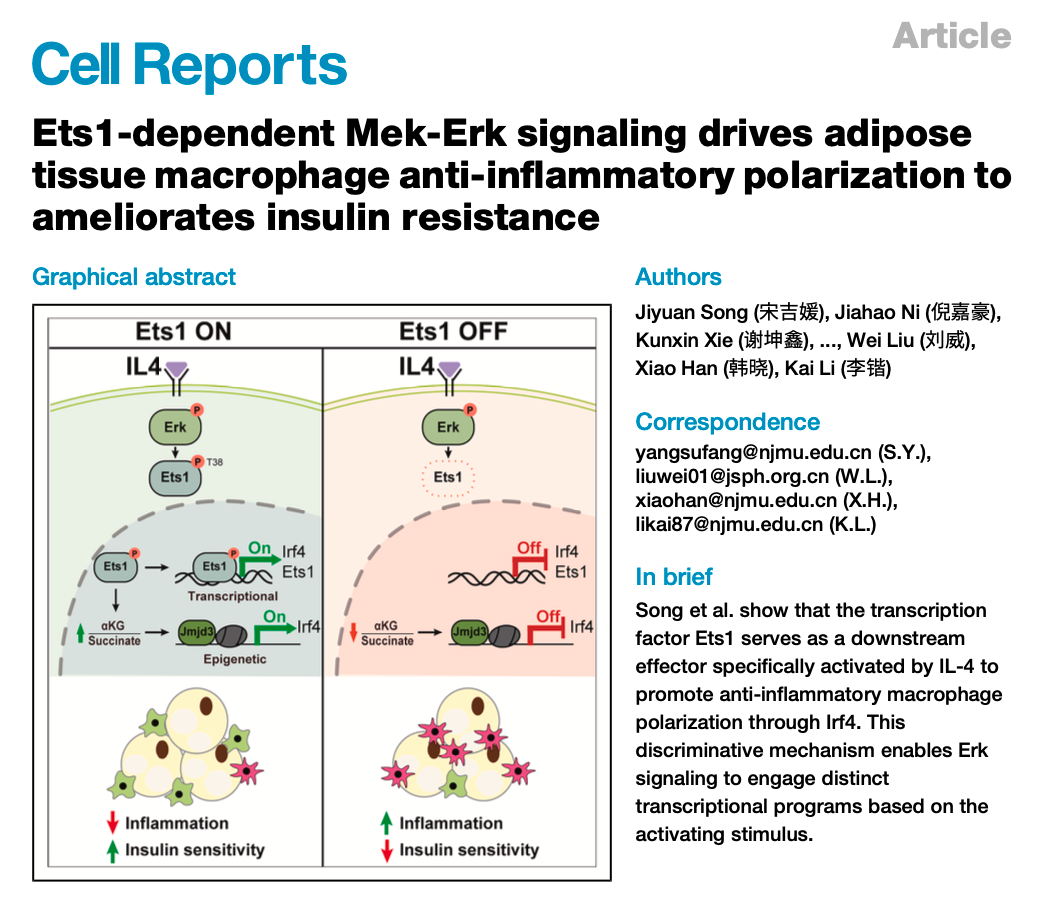
In their study, the team compared the effects of IL-4 (an M2 polarization inducer) with LPS/palmitic acid (M1 polarization inducers) on macrophages. They found that although all stimuli activated Erk phosphorylation, only IL-4 significantly upregulated Ets1 protein levels in a Mek-Erk signaling-dependent manner. This finding indicates that Ets1 servesas a differentially regulated node in the Mek-Erk pathway, responding specifically to upstream signals via Mek-Erk-mediated anti-inflammatory signals. Using myeloid-specific Ets1 knockout mice and other animal models, the team further confirmed that Ets1 deficiency exacerbates high-fat diet-induced adipose tissue inflammation and insulin resistance. Mechanistically, through multi-omics screening and validation, the study established that Ets1 drives anti-inflammatory polarization through a dual mechanism: on the one hand, Ets1 directly binds to the Irf4 promoter region to promote its transcription; on the other hand, Ets1 transcriptionally represses the Ogdh gene, increasing the α-ketoglutarate/succinate ratio, thereby activating JMJD3-mediated epigenetic remodeling and enhancing Irf4 expression. Finally, macrophage transplantation experiments confirmed that replenishing Irf4 can reversethe local adipose inflammation and insulin signaling impairment caused by Ets1 deficiency.
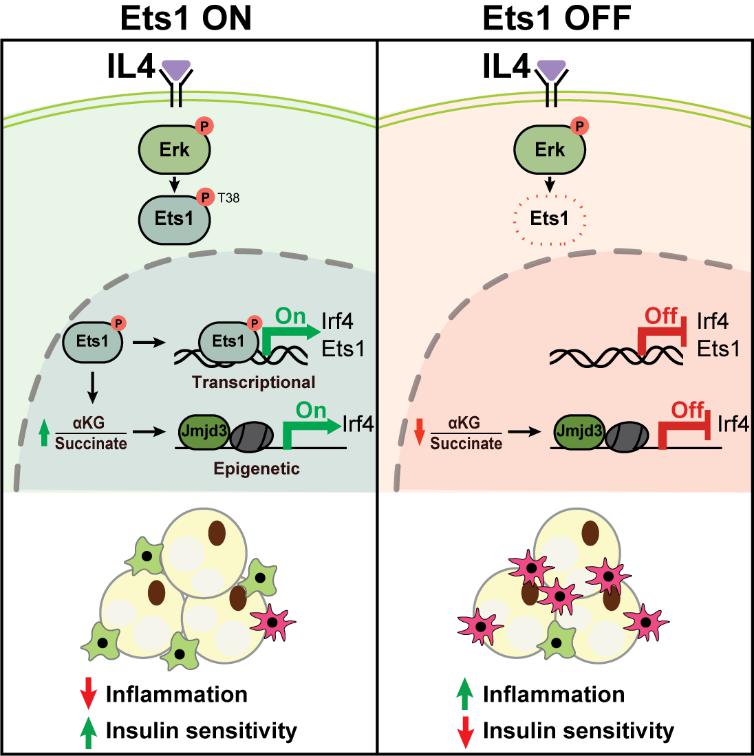
In summary, this study identifies a key regulatory mechanism in the Mek-Erk signaling pathway: Erk drives anti-inflammatory polarization of macrophages by activating the specific downstream effector Ets1, thereby alleviating adipose tissue inflammation and insulin resistance. This provides a new perspective for understanding the immune-metabolic regulatory function of Erk signaling and offers a potential intervention target for obesity-related metabolic diseases.
Li Kai and Han Xiao from the School of Basic Medical Sciences at NMU, Liu Wei from the First Affiliated Hospital with NMU, and Yang Shufang from the Affiliated Taizhou People’s Hospital of NMU are co-corresponding authors. Graduate students Song Jiyuan, Ni Jiahao, and undergraduate student Xie Kunxin from the School of Basic Medical Sciences, NMU are co-first authors. The research was supported by grants from the National Natural Science Foundation of China and other funding sources.
Original link: https://doi.org/10.1016/j.celrep.2026.112744
(Drafted by the research team led by Li Kai; Reviewed by Wang Juejin; Translation revised by Bei Zhang)



