Recently, Professor Zhipeng Xu and Professor Minjun Ji’s research team from Nanjing Medical University (NMU), in collaboration with Professor Zikai Zhou’s team from the Shanghai Institute of Materia Medica, Chinese Academy of Sciences, published agroundbreaking study in Cell Reports. The paper, titled “Maternal immune activation perturbs intestinal niche through microbial glycerophospholipids and drives offspring behavioral abnormalities”,offers a novel insight into the molecular mechanism by which maternal immune activation (MIA) during pregnancy, via the gut microbiota metabolite 1-oleoyl-2-myristoyl-sn-glycero-3-phosphocholine (OMPC), leads to autism spectrum disorder (ASD)-related behavioral abnormalities in offspring. Furthermore, the study proposes that Lacticaseibacillus rhamnosus GG (LGG), when administered as a prenatal preventive strategy, can effectively alleviate placental and fetal neuroinflammation induced by MIA.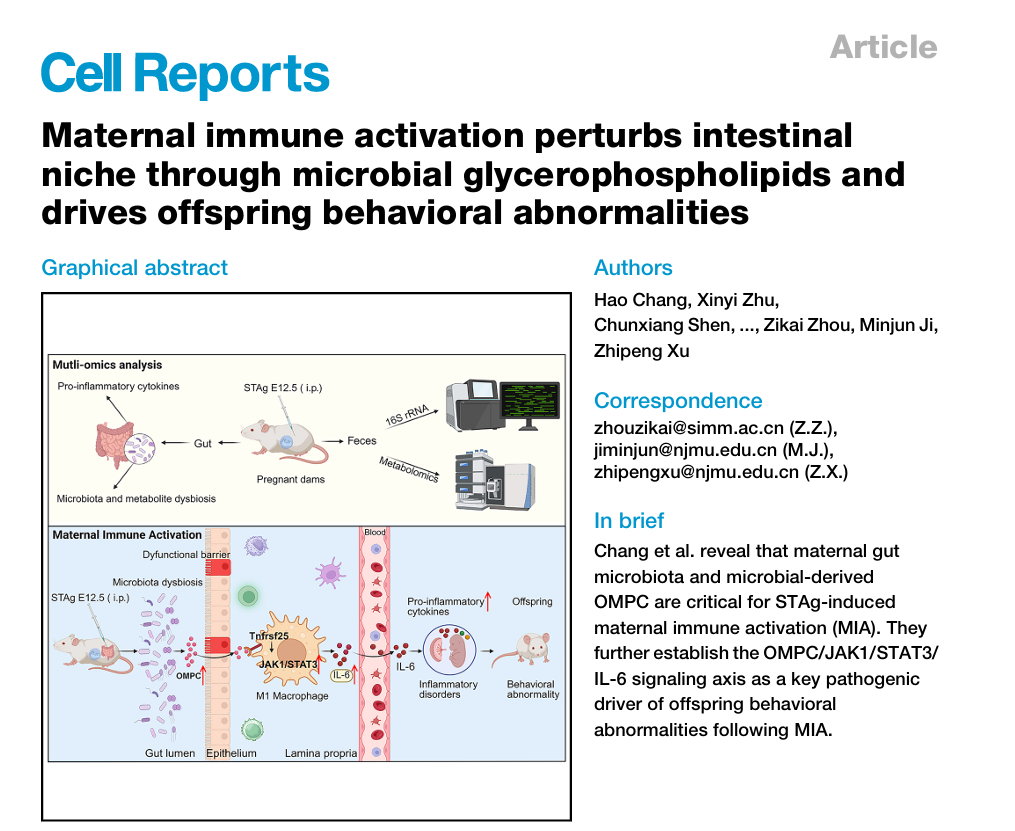
Autism spectrum disorder (ASD) is a neurodevelopmental disorder (NDD) resulting from the complex interplay of genetic and environmental factors. ASDsignificatnly affects cognition, motor function, social adaptation, and behavior, severely impeding the growth, development, and mental well-being of affected children. Maternal immune activation (MIA), which occurs due to maternal infection during pregnancy, has been identified as a key factor in the development of ASD in offspring. Therefore, gaining a deeper understanding of the molecular network through which maternal immune status during pregnancy influences autism pathogenesis in offspring, along with identifying effective early intervention targets and strategies, is crucial for promoting children’s mental health and preventing the onset of major neurodevelopmental disorders at their root. .
Building upon a previously established MIA mouse model induced by soluble tachyzoite antigen (STAg) of Toxoplasma gondii (Nature Neuroscience, 2021), the research team employed a multi-omics approach to comprehensively study the pathological progression of the gut, placenta, and fetal brain in STAg-MIA pregnant mice. Key findings include a markedly enhanced pro-inflammatory response in the gut of MIA dams, characterized by M1 macrophage polarization and significant upregulation of the key pro-inflammatory cytokine IL-6. Throughintegrating gut microbiota and metabolomic profiling, the study revealed significant gut dysbiosis and pronounced metabolic perturbations in STAg-MIA dams, including a significant increasein the pro-inflammatory metabolite OMPC. This elevation led to a marked upregulation of pro-inflammatory phenotypes in both the placenta and fetal brain, ultimately contributing to behavioral abnormalities in offspring. Moreover, the study confirmed that maternal IL-6 signaling plays a pivotal role in mediating offspring behavioral abnormalities induced by STAg-induced MIA. Both IL-6 neutralizing antibody treatment and IL-6 genetic knockout significantly alleviated STAg-MIA-induced placental inflammation and fetal neuroinflammation. Offspring behavioral assessments showed that maternal IL-6 blockade significantly ameliorated core autistic symptoms, including deficits in social interaction and repetitive stereotyped behaviors.
Additionally, Spearman correlation analysis revealed a strong positive correlation between OMPC levels and the upregulation of intestinal IL-6 expression. Mechanistically, this study demonstrates that OMPC binds to the Tnfrsf25 receptor on the macrophage surface, activating the JAK1/STAT3/IL-6 pro-inflammatory signaling axis. This activation leads to significant increase in IL-6 levels in both the maternal gut and peripheral blood, ultimately resulting in offspring behavioral abnormalities.
Based on these mechanistic insights, the research team further explored potential intervention strategies targeting the gut microenvironment. Given the increased intestinal inflammation in STAg-MIA pregnant mice, the team administered oral supplementation withLacticaseibacillus rhamnosus GG (LGG), an anti-inflammatory probiotic, during early pregnancy. The results showed that LGG intervention effectively suppressed maternal intestinal IL-6 expression, restored gut barrier integrity, and significantly alleviated the inflammatory response in both the placenta and fetal brain of STAg-MIA dams. Consequently, this intervention prevented the development of behavioral abnormalities in the offspring. This finding offers a promising preclinical approach for early intervention in MIA-associated neurodevelopmental disorders.
In summary, this study, for the first time, defines OMPC/JAK1/STAT3/IL-6 signaling pathway as a pathogenic hub in MIA. Moreover, it highlights LGG as a promising prenatal prophylactic strategy to mitigate neurodevelopmental disorders.
Original link:
https://www.cell.com/cell-reports/fulltext/S2211-1247(26)00289-5
(Drafted by ZhipengXu, Minjun Ji, and Zikai Zhou’s Research Team; Reviewed by Juejin Wang; Translation revised by Bei Zhang)



