Recently, AA research team led by Professor Xiang Zhou from the Cardiovascular Center of the Second Affiliated Hospital with Nanjing Medical University (NMU) recently published a paper entitled “METTL3-mediated N6-methyladenosine modification of Dnajb1 modulates cardiomyocyte ferroptosis during myocardial infarction” in the prominenestigioust international journal PNAS. For the first time, theThis study,for the first time,elucidateds the regulatory mechanisms of the METTL3/Dnajb1/GPX4 axis in myocardial infarction (MI).

MIAs a is a leading major cardiovascular disease, thatMI poses a severeignificant threat to human health. During the pathological injury process induced by myocardial ischemia and hypoxia, massive cardiomyocyte death occurs, which can directly lead to a sharp decline in heart function, ultimately resulting ininducing heart failure. In recent years, Fferroptosis, is a novel form of programmed cell death characterized by iron dependence and lipid peroxidation. In recent years, it has become, has emerged as a major research hotspotfocus in the field of cardiovascular diseases. Therefore, elucidatingunderstanding the molecular mechanisms of cardiomyocyte ferroptosis is crucial for reducing cell death, attenuamitigating adverse cardiac remodeling, and preventing post-MI heart failure.
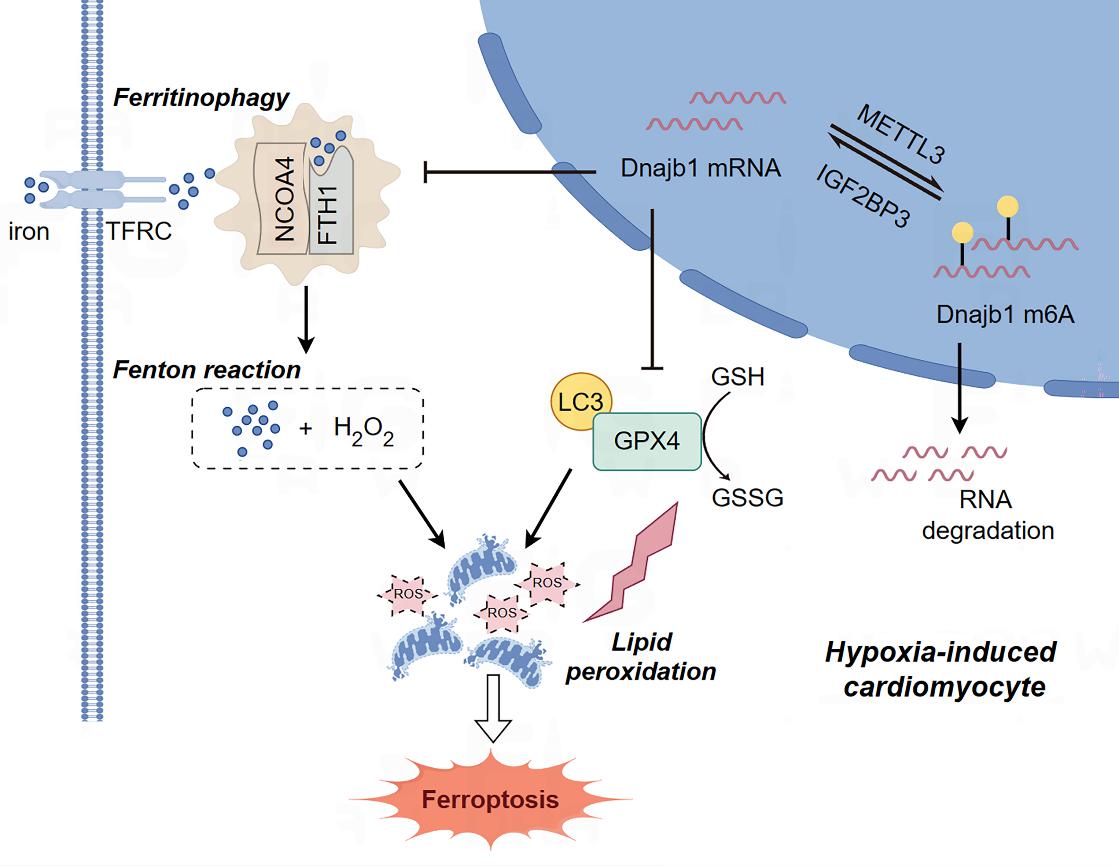
This study By conducting an integrated analysis of methylated RNA immunoprecipitation-sequencing and RNA-sequencing data obtained from the myocardial tissues of MI mice, and the researchers identified DNAJB1 as a key gene that exhibitinged increased N6-methyladenosine (m6A) modification and decreased mRNA abundance duringin the context of ischemic myocardial injury. Further researchinvestigation revealed that MI-induced upregulation of METTL3 decreasereduces Dnajb1 mRNA stability through m6A modification, which leadings to the lysosomal degradation of the core protein GPX4, thereby driving cardiomyocyte ferroptosis. These findings revealhighlight the crucial regulatory role of m6A epigenetic modifications in ischemic myocardial injury, openingproviding new avenueinsights for the futuredevelopment of targeted therapy of for myocardial infarction.
Professor Xiang Zhou is the corresponding author of this paper, with Shuchen Zhang, Boyang Xiang, and Yiheng Zhao serving as co-first authors; a. All researchers are affiliated with the Second Affiliated Hospital of our universitywith NMU.
原文链接Original article link:https://doi.org/10.1073/pnas.2529187123
(Translation revised by Bei Zhang)



